GLUCAGON-LIKE PEPTIDE-1 (GLP-1)
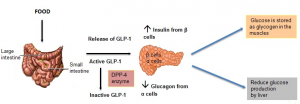
Glucagon-like peptide-1 (GLP-1) is a hormone that is released from the intestines (ileum and colon). The secretion of GLP-1 is depending on the presence of nutrients in the small intestines.
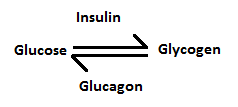
As we know, insulin is a hormone that helps to control the blood sugar (glucose) level.
- It is secreted from the ß-cells in the pancreas.
- When the blood sugar is high in the intestines, GLP-1 is released1-5 and this stimulates the secretion of insulin.
- Insulin stimulates the uptake of glucose into the muscles, liver and fat cells so that the glucose level is maintained within normal limits in the blood.
- Glucose is then stored as glycogen in the muscle, liver and fat tissues.
GLP-1 also inhibits the secretion of glucagon from the ?-cells in the pancreas. Glucagon is a hormone that stimulates the release of glucose from the liver and muscle when the blood sugar level is low. With the presence of GLP-1, the secretion of glucagon is suppressed and the level of sugar in the blood is reduced1-5.
Since the secretion of GLP-1 is glucose-dependent, when the concentration of sugar is normal in the blood, GLP-1 will not be released6. Therefore, low blood sugar problem (hypoglycaemia) can be avoided.
GLP-1 may help to preserve the ß-cell function and helped to increase the insulin in the body. It inhibits the ß-cell death and helps to increase the ß-cell in the body5. In addition, GLP-1 also inhibits gastric emptying. This delays the absorption of carbohydrates in the intestines7. As the food stays longer in the intestines, it contributes to the feeling of fullness and reduces food intake. When the intake of food is low, one may lose weight.
However, GLP-1 is only active in the body for less than 2 minutes as it quickly deactivated by an enzyme called dipeptidyl peptidase-IV (DPP-IV)8. In diabetes patients, GLP-1 that is secreted by the gut is reduced thus resulting in impaired insulin secretion and the release of glucagon is also not suppressed. Following these, the blood sugar is high in diabetes patients9.
Since GLP-1 has multiple benefits, it has the potential for treatment in diabetes. In order to increase the GLP-1 in the body, we can either supplement with external GLP-1 or inhibit the deactivation of this hormone.
The first artificial GLP-1 that is introduced to the market for the treatment is type 2 diabetes is exenatide (Byetta®). Exenatide is derived from the hormone found in the saliva of a lizard called Gila monster.
- It is given as injection under the skin at the fatty area (subcutaneous) of the abdomen, thigh or arm as it is rapidly deactivated by gastric acid if given by mouth.
- The initial dose is 5mcg to be injected twice a day, anytime within 60 minutes of the first meal and last meal of the day10. The gap between the two injections shall be six or more hours apart.
- After one month, your doctor may increase the dose to 10mcg to be injected twice a day.
- It has to be kept in the refrigerator11.
- The difference between exenatide and GLP-1 is exenatide is not deactivated by the DPP-IV enzyme in the body. As the result, exenatide stays longer in the body than the GLP-1.
Another artificial GLP-1 is liraglutide marketed under the brand Victoza®. Liraglutide is also given as subcutaneous injection but it is once daily injection.
- The starting dose is 0.6mg daily for one week. The starting dose is low to be effective but it will reduce the side effects of nausea and vomiting.
- After one week, the doctor may increase the dose to 1.2mg daily.
- If your blood sugar is not adequately control, the doctor may further increase the dose to 1.8mg daily.
- It can be injected at anytime of the day and preferably at the same time every day.
- Liraglutide has a fatty acid molecule that binds to the protein (albumin) in the body, thus making it stays longer in the body12.
- It can be placed at room temperature and lasted for 28 days.
Artificial GLP-1 is indicated as an additional treatment of type 2 diabetes patients that still do not achieve good glucose control with metformin alone or other combination therapies of metformin with sulphonyurea or thiazolidinedione. It shall be used before commencing insulin therapy. Possible benefit of starting this therapy earlier is that it may help to reduce blood sugar without causing hypoglycaemia and may reduce body weight as well as preserve the ß-cells.
References
- Nauck MA, Homberger E, Siegel EG, Allen RC, Eaton RP, Ebert R, et al. Incretin effects of increasing glucose loads in man calculated from venous insulin and C-peptide responses. J Clin Endocrinol Metab. 1986;63:492-498.
- Drucker DJ. Enhancing incretin action for the treatment of type 2 diabetes. Diabetes Care. 2003; 26:2929-2940.
- Drucker DJ. Incretin-based therapies: a clinical need filled by unique metabolic effects. Diabetes Educ. 2006;32(suppl 2);65S-71S.
- Visboll T, Holst JJ. Incretins, insulin secretion and type 2 diabetes mellitus [review]. Diabetologia. 2004;47:357-366.
- Drucker DJ. Glucagon-like peptide-1 and islet ?-cell: augmentation of cell proliferation and inhibition of apoptosis [review]. Endocrinology. 2003;144:5145-5148.
- Nauck MA, Kleine N, Orskov C, Holst JJ, Willms B, Creutzfeldt W. Normalization of fasting hyperglycaemia by exogenous glucagon-like peptide 1 (7-36 amide) in type 2 (non-insulin-dependent) diabetic patients. Diabetologia 1993. 36:741-744.
- Gutzwiller JP, Drewe J, Goke B, Schmidt H, Rohrer B, Lareida J, et al. Glucagon-like peptide-1 promotes satiety and reduces food intake in patients with diabetes mellitus type 2. Am J Physiol. 1999;276(5 Pt 2):R1541-R1544.
- Mentlein R. Dipeptidyl-peptidase IV (CD26)-role in the inactivation of regulatory peptides [review]. Regul Pept. 1999;85:9-24.
- Muller WA, Faloona GR, Aquilar-Parada E, Unger RH. Abnormal alpha-cell function in diabetes. Response to carbohydrate and protein ingestion. N Engl J Med. 1970;283:109-115.
- Byetta package insert Accessed September 6, 2012
- http://www.medicinenet.com/exenatide/article.htm, Exenatide, accessed September 5, 2012
- http://www.medicinenet.com/liraglutide/article.htm, Liraglutide, accessed September 5, 2012
 PENDIDIKAN PESAKIT Kementerian Kesihatan Malaysia
PENDIDIKAN PESAKIT Kementerian Kesihatan Malaysia